Quick Menu
Sulfuric Acid Monitoring
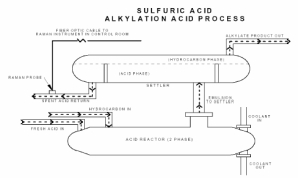
A need exists for a reliable, in situ method to continually monitor sulfuric acid concentration in the alkylation process. Ideally, a noncontact sensor would be inserted into the acid process in an appropriate location and allowed to communicate with a base instrument located remotely in the safe environment of a control room. Process Instruments Inc. has started developing a fiber coupled, Raman scattering instrumentation for this application. The fiberoptic Raman probe is manufactured out of carbon steel and can be directly inserted into the process stream with no sample conditioning required.
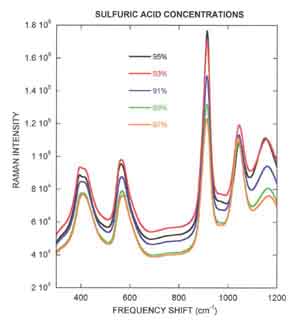
Process Instruments PI-200-L instrument was used to record the Raman spectra of l8 laboratory prepared sulfuric acid dilutions ranging from 95% to 85% acid mixed with water. The figure to the right illustrates how the Raman spectra change with acid concentration. The Raman peaks located at 913 cm-1 and 1162 cm-1 both decrease when the acid is diluted while the Raman peak at 1043 cm-1 increases substantially. The fact that some peaks decrease with decreasing acid concentration and others increase create a very robust chemometric model that is used to correlate the Raman spectra to acid concentrations.
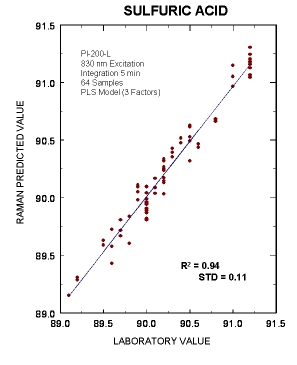
Partial Least Squares analysis of the percent sulfuric acid concentration from 64 alkylation unit samples. The Raman spectrum of each alkylation unit sample along with the laboratory titration measurement were used to make the PLS prediction. The PLS model required three individual factors, and these factors correspond to the three acid peaks in the 850 to 1250 wavenumber region. The standard deviations of the (predicted minus laboratory) values was 0.11 %Wt, and this variance is within the 0.2%Wt that is inherent in the conventional titration technique. The observable agreement between the predicted and actual sulfuric acid concentration for the various alkylation unit samples demonstrates that on-line Raman spectroscopy can be utilized for in-situ sulfuric acid monitor.
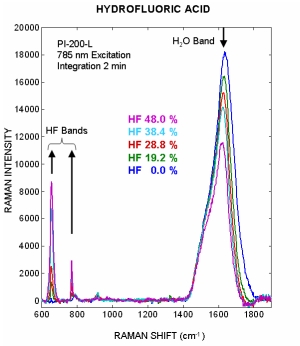
Hydrofluoric Acid Monitoring
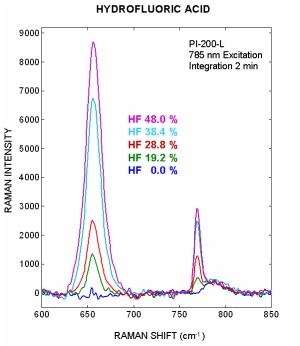
Process Instruments PI-200-L instrument was used to record the Raman spectra of 4 laboratory prepared hydrofluoric acid dilutions ranging from 48% to 19.2% acid mixed with water. The figure to the right illustrates how the main HF acid peaks located at 655 cm-1 and 759 cm-1 decrease in intensity when the acid is diluted with water. Hydrofluoric alkylations units require a much higher acid concentration then we could obtain for this study. Since both of the main HF peaks increase with concentration the elevated alkylation HF acid concentrations will produce an improvement in the signal to noise ratio when compared to this initial study.